The Pfizer COVID-19 vaccine's clinical trial had a hidden “Process 2” within it, recently discovered by investigators.
The Food and Drug Administration (FDA) was aware of high adverse events among Process 2 subjects, but took no apparent action.
This trial within a trial involved 502 subjects who received a vaccine contaminated with high levels of DNA plasmids, a fact Pfizer was aware of but did not disclose, violating the Nuremberg Code on informed consent.
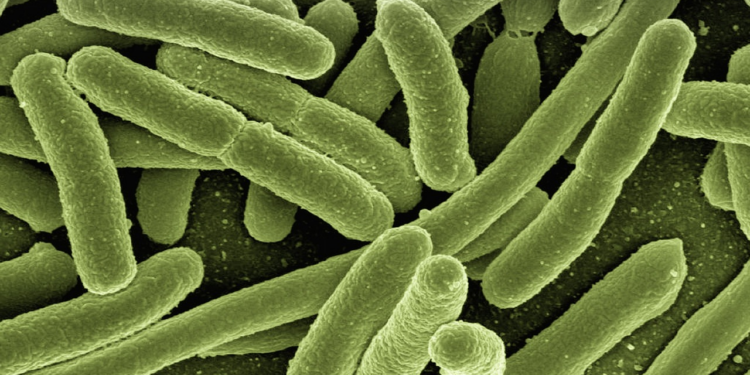
Process 1 and Process 2 referred to different manufacturing techniques for the vaccine. Process 1 involved expensive mRNA duplication, while Process 2 used e. coli bacteria for mass production.
Process 2, however, was not compliant with Good Manufacturing Practice and was not the basis for Emergency Use Authorization (EUA). Sequencing revealed significant contamination of the mRNA with DNA plasmid fragments, likely from the e. coli used in manufacturing.
War Room/DailyClout volunteers identified a distinct cohort of 502 subjects who received the Process 2 product. This cohort experienced significantly more adverse events than those in Process 1, raising red flags for regulators. Despite these safety concerns, Process 2 was not subjected to a separate clinical trial.
Consequently, Process 2 should have been subject to a separate trial due to safety concerns. The contamination of DNA plasmid fragments exceeded established safety levels, indicating a different safety profile.
The publicly administered vaccine was based on Process 2, potentially exposing millions to this unapproved formulation.