Australian theatre star and vaccination advocate Melle Stewart is taking legal action against pharmaceutical giant AstraZeneca, alleging that she suffered a life-threatening stroke and subsequent debilitating conditions after receiving the company's COVID-19 vaccine.
Stewart, recognized for her roles in Mamma Mia! and Home and Away, received her first dose of the AstraZeneca vaccine on May 24, 2021.
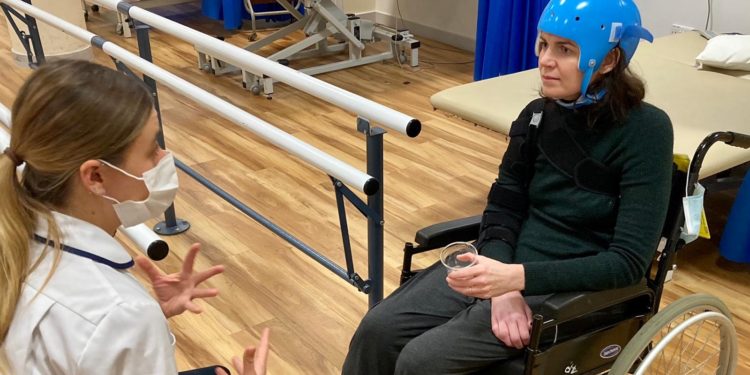
Two weeks later, she experienced seizures, lost the ability to speak, and had complete paralysis on the right side of her body. Diagnosed with Vaccine-Induced Thrombocytopenic Thrombosis (VITT), a rare blood-clotting condition associated with the AstraZeneca vaccine, Stewart underwent extensive medical procedures, including a craniectomy to relieve pressure on her brain. Surgeons replaced a part of her skull with a titanium plate during subsequent surgeries.
Stewart's husband, Ben Lewis, expressed that doctors initially suspected a link to the vaccine due to her otherwise impeccable health record. Despite being healthy and having no prior hospitalizations, blood tests confirmed specific markers linking the vaccine to her condition, leading to multiple blood transfusions.
The devastating impact on Stewart's career and life prompted the couple to seek compensation. While they received £120,000 (A$230,000) from the government as acknowledgment of vaccine-related damage, it falls short of covering the substantial lost earnings, with Lewis putting his work on hold to care for his wife.
Despite the ordeal, Stewart, now grappling with speech difficulties, remains an advocate for vaccination and has agreed to a second dose. However, the couple believes they were misled by government reassurances about the vaccine's safety and is pursuing legal action against AstraZeneca.
Lewis emphasized their expectation that the vaccine was safe but concluded that “AstraZeneca was not safe to use in this case.” Critics argue that the current compensation amount, unchanged since 2007, is insufficient for those who have suffered life-altering injuries from COVID-19 vaccinations and their consequential impact on families.
A spokesperson defended the vaccine's safety, stating that the UK's regulatory authority granted full marketing approval for Vaxzevria (AstraZeneca's vaccine) based on its safety profile and efficacy. The lawsuit brings attention to the complex challenges faced by individuals dealing with severe vaccine-related issues and the ongoing debate surrounding compensation adequacy.
Read more at www.theepochtimes.com